Decolonising Dermatology: Why Black and Brown Skin Need Better Treatment
Neil Singh, The Guardian, August 13, 2020
As I sat making small talk with my patient, I was trying not to think about the ants. It was “flying ant day”: that moment every summer when the heat inspires thousands of ants to grow wings and swarm in their thousands, mating mid-air in a huge, frenzied orgy. My only source of cool air was a skylight, but having it open meant that every so often stray black garden ants were dropping between me and my patients, making me jumpy.
My patient was a rangy black man, early 40s, with hair just starting to grey at the temples. He looked younger than he was, wearing a black T-shirt and cut-off denim shorts. It was well into music festival season, and he wore several wristbands to prove it. I asked what kind of music he was into. It turned out he was not just a music fan; he was a roadie and gig manager. He had come in to ask me about his skin. He lifted up his T-shirt, showing me his chest, and I was shocked to see a splash of rough, silvery patches all over his torso.
I had no idea what to make of them. I was initially quite alarmed, thinking of a host of possible causes, some really serious. But he reassured me: “I get these quite a lot.” He saw my blank look and explained, politely. “I have psoriasis.” I went back to his medical record, and there it was, a listed diagnosis that I had failed to spot: chronic plaque psoriasis. He was having a flare-up and wondered if there was an alternative treatment he could try. In the end, it was a straightforward case, which simply required a trial of a different cream. But, after he’d gone, I was left feeling unsettled, because if this had been the first time he’d had this rash, I would probably have been way off the mark with my diagnosis.
I thought back to the dermatology training I had received in medical school. I could recall only three occasions when dark skin was used to demonstrate a skin problem: once, to illustrate how black skin can heal in thick bands called keloid scars; once, during a session about syphilis and other genital ulcers; and once, in a presentation on vitiligo, a disorder in which patches of skin lose their pigment. Dark skin had only ever been used to demonstrate these dermatological rarities, and never as part of core teaching on common disorders.
I am not the only one reflecting on this failure of medical education. In June, Michael Mackley, a third-year medical student in Canada, wrote a widely shared Twitter thread about feeling unprepared to spot skin changes in a black patient. “Patients are left seeking equal care in a system that is entirely built on white skin,” he wrote. But what made my own ignorance all the more unsettling was that this had never occurred to me as a problem, even though I am a person of colour.
At a Black Lives Matter protest in Brighton this summer, I saw a black teenager proudly wearing a T-shirt bearing the word “MELANINAIRE”. This brand name is eye-catching precisely because, in nearly every other circumstance, the amount of melanin in your skin is directly proportional to the amount of discrimination you encounter in everyday life. After all, melanin has been used, along with facial features and hair type, for more than four centuries to sort the world into a hierarchy, with white-skinned groups at the top.
Melanin – which comes from the Greek word, melas, meaning dark – is a group of pigments found throughout the natural world, responsible for the brown spots on bananas, the ink of cephalopods such as squid, and even the colour of ants. Three types of melanin are found in the human body, but the most common is eumelanin: a brown-black pigment responsible for most of the colouring of human skin, hair and eyes. It is made in specialised skin cells, melanocytes, where it is packaged into discs called melanosomes, which are passed on to other skin cells to help protect our DNA from sun damage. These discs surround the nucleus, guarding it like tiny parasols, blocking out ultraviolet rays.
The difference between light and dark skin is simply the amount and distribution of eumelanin in cells in the epidermis, the outermost layer of skin. That’s it. Although we can temporarily raise the amount of eumelanin in our skin by tanning, the baseline amount is controlled by our genes. Which raises the question: why don’t all humans have the same colour skin?
The best answer we have has been offered by Nina Jablonski, a professor of anthropology at Penn State University, and author of Living Color: The Biological and Social Meaning of Skin Color. Her research has shown that this baseline level of eumelanin has evolved differently in different populations, based on the need to optimise the amount of two essential vitamins: folate and vitamin D. Folate is used to regenerate fast-growing tissues, such as those in sperm and embryos. Without sufficient folate, men can become infertile and babies can develop spinal cord defects. Meanwhile, vitamin D is required to absorb calcium from our diet. Without sufficient vitamin D, we cannot form normal skeletons or immune systems.
The ultraviolet radiation in sunlight depletes the level of folate in your body, which is why eumelanin’s ability to block such radiation is so useful. But there’s a catch. Ultraviolet radiation is also vital: vitamin D cannot be made without it. To survive and reproduce, our skin needs to allow in just enough ultraviolet radiation so we can produce sufficient vitamin D, but not so much that our folate levels get dangerously low. Nearer the equator, there is more ultraviolet radiation to block, which is why humans whose ancestors evolved in this region have more eumelanin. As some human populations migrated nearer the poles, where there is less ultraviolet radiation reaching the skin, humans in these regions evolved to have less eumelanin (and thus paler skin), to prevent vitamin D deficiency.
Skin colour became just one of many genetic divergences that occurred between geographically separated populations over time; no more socially relevant than other genetic bifurcations that could have been used to divide populations – such as whether you can or can’t digest milk, or whether you have wet or dry ear wax. For millennia, skin colour meant difference, but not systematically differential treatment. But in Europe, during the early modern period, skin colour began to take on a very different meaning. In an age of rising capitalism and colonialism, it became a convenient way for white people to divide up the world into those who would labour hardest, and those who would benefit most from the wealth this produced. Irene Silverblatt, a professor of cultural anthropology at Duke University, describes how the Spanish were the first to systematically divide the world by skin colour in the 17th century: “white, black, and brown [became] abridged, abstracted versions of coloniser, slave, and colonised.”
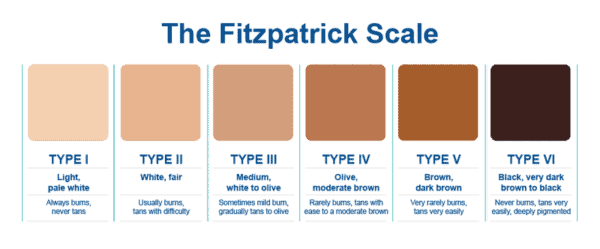
These days, skin colour is formally categorised into one of six gradations called the Fitzpatrick phototypes. These range from I (pale skin that never tans and burns easily) to VI (dark skin that never burns). This schema is named after Thomas B Fitzpatrick, an influential white dermatologist who worked at Massachusetts General hospital in the second half of the 20th century. A wise-cracking, pipe-smoking detective of dermatology who kept a magnifying glass in his watch pocket, Fitzpatrick formed a double-act with Wallace H Clark, his more introverted pathologist colleague. For several decades, they were the Holmes and Watson of skin cancer: Fitzpatrick researched how healthy tanning can go awry and cause cancer, while Clark developed a staging system of skin cancer depth that is still used today.
But Fitzpatrick’s spectrum reflects his own biases as much as it does objective science. His phototypes are skewed towards white people, with three of the six types covering what we would commonly call white skin, two for brown skin, and only one type to encompass the many kinds of blackness.
In recent years, a generation of race-conscious medical students who are used to choosing the exact skin colour of their emojis have raised concerns that the images through which they study are almost exclusively white. From the anatomy posters they hang above their beds, to the plastic mannequins they practise chest compressions upon, the default patient is almost always the same: a white-skinned man (only a woman when showing a relevant organ) with a hairless body and a neat side-parting of trimmed, mousy hair. This persists, despite the fact that white people are a global minority.
A 2018 study analysed more than 4,000 images in four major US medical textbooks, and found that only 4.5% of images showed dark skin. This is a problem in every branch of medicine, as it reinforces whiteness as the norm, but it is most troubling in the teaching of dermatology, where every problem is set against the canvas of skin it arises from. Ademide Adelekun and Ginikanwa Onyekaba, from the Perelman School of Medicine at the University of Pennsylvania, recently published a similar study, but this time focusing on dermatology textbooks. Only 11.5% of images showed dark skin (of Fitzpatrick phototype V or VI). Earlier this year, I gave a lecture at the medical school I work at, about common skin problems. Even making a conscious effort to include pictures of various skin tones, I found it difficult to find relevant images in online medical libraries.
“Not all patients present with illness in the same way,” said Subash Jayakumar, in a lecture he gave in 2018 at University College London Medical School, where he teaches. “Significant differences exist in patients from different ethnic backgrounds.” Jayakumar works as a primary care physician in Brent, a London borough where 65% of residents are people of colour. He reminded the audience that skin changes are easy to miss if you don’t know what you are looking for. Blue-tinged fingertips are a harbinger of dangerously low oxygen levels; the yellow stain of jaundice can be an early warning sign of irreversible liver damage; and a patient who is bleeding internally might be saved by a doctor who knows to look for pallor not in the cheeks, but under the eyes and inside the mouth. How can we expect doctors to correctly name a colour change in coloured skin if they’ve never seen it before?
This reminded me of a problem that the photography world has been grappling with for decades. Just like in medicine, modern photography was designed with white people in mind. In fact, a white person with a name: Shirley. From the 50s, Shirley was the name given to the woman on the test cards against which all photographic laboratory technicians would judge their colour balance and exposure when using Kodak film. The model chosen for the test card changed with each decade, but the formula was always the same: an ivory-skinned brunette who represented “normal”, allowing every technician to develop all Kodak film in a way that made Shirley look ideal.
The problem was that it made people of colour look terrible: erasing the subtleties of tone and contour, so their faces became black blobs with the bright whites of their eyes and teeth glaring out. In 1977, the French-Swiss film director Jean-Luc Godard refused to use Kodak film when filming in Mozambique, calling it “racist”. It was only in the 80s, after manufacturers of chocolate and wooden furniture had complained that their products all looked the same using their film, that Kodak developed new film stocks. Kodak introduced Gold Max film, boasting that it would allow any photographer “to photograph the details of a dark horse in low light”.
The problem still persists, to a lesser degree, in digital photography. Despite improvements in colour-balancing and colour-correction, Sarah Lewis, an assistant professor at Harvard university, writes that “if the light source is artificial, digital technology will still struggle with darker skin”.
Medicine seems to have been built with the same solipsism as photography. For instance, pulse oximeters – those devices health workers peg to your finger, which read your oxygen levels by pinging a red light through your skin – were calibrated using white patients, and they have been shown to consistently overestimate oxygen levels in dark-skinned people by up to 7%. “If we don’t fix the biases we have in healthcare before we jump to training a computer to do it, we’re just going to actually perpetuate those biases,” said Roxana Daneshjou, a dermatologist at Stanford University, in an interview with National Public Radio last year.
There are several artificial-intelligence smartphone apps that claim to be able to identify skin conditions. One of these apps, Skin Image Search, compares the image you upload to a database in which less than 10% of images show dark skin. Ugandan researchers have reported that the diagnostic accuracy of this app was just 17% for dark skin when they tested it on a set of 123 patients with black skin. Here we see how the failings in these disciplines interconnect: even if medical textbooks and apps do become based on a proportionate number of pictures of patients of colour, the images are less likely to be accurate if cameras aren’t able to properly capture richly pigmented skin.
Still reeling from my own inability to recognise psoriasis in the dark skin of my patient, I spoke to other patients of colour who had bad experiences seeking treatment for skin problems. Afia Ahmed Chaudhry, a history teacher and researcher based in London, told me how her mother felt her skin colour meant she got stuck in a cycle of mistreatment. “[Patients of colour] are often fobbed off,” Chaudhry told me, “and then you keep going round and round until you give up, or have to pay to go private.” She is furious that it took her mother 15 years to be offered the correct treatment for her severe acne. “She shouldn’t have had to wait so long,” she said. “They shouldn’t have taken advantage of her and given her the same topical treatments that never worked.”
For centuries, African societies have had expertise in dealing with richly pigmented skin, using infusions, poultices and ointments made from plants: from the prickly pear extracts used as emollients, to aloe vera used to heal wounds, to rooibos used to treat dermatitis. But much of this traditional knowledge has been lost. Mohamed Soumah – a professor of dermatology at Donka National hospital, in Conakry, Guinea – told me that “in Africa there is little work that deals with dermatology on dark skin”. African doctors still study using textbooks mainly written in the west, he said, so doctors must pick up what their books fail to teach them through everyday exposure to dark-skinned patients.
To address western medicine’s ignorance of patients of colour, clinics have been set up where people can see dermatologists who take a special interest in darker skin. Most major western cities now have such “skin of colour clinics”, many operating under the banner of “ethnic dermatology” or “multicultural dermatology”. The first dedicated skin of colour clinic was created by John A Kenney Jr, who was born in Alabama in 1914. His father was a pioneering black surgeon who became known for his social activism; he wrote a book called The Negro in Medicine, and tried to persuade local hospitals to employ more black doctors. One night in 1922, when Kenney Jr was only eight years old, he and his family woke to find the Klu Klux Klan had burned a cross on their front lawn. After repeated death threats, one delivered by a patient of Kenney’s father while he was treating him, the family eventually fled to New Jersey.
Kenney went on to become one of the first black dermatologists in the US, setting up the first skin clinic specifically aimed at black people, at Howard University hospital, just outside Washington DC. Kenney died in 2003, but his legacy remains; by the time of his death, he had personally trained a third of the black dermatologists in the US.
The premise behind having such dedicated skin of colour clinics is that dark skin is, in a clinically relevant way, different to white skin. But exactly what and how medically important these differences are is still contested. In many cases, long-debunked ideas remain widespread. In 1851, Samuel Cartwright, a pro-slavery doctor, argued that black people had thicker, less sensitive skin, as part of a disorder he called “dysaesthesia aethiopica”, which he thought made workers sluggish. He taught that the only cure was a good whipping. “The best means to stimulate the skin,” he wrote, is “to anoint it all over in oil, and to slap the oil in with a broad leather strap; then to put the patient to some hard kind of work in the sunshine.” A 2016 study showed that a third of American medics still believe myths about black patients, such as thinking they have thicker skin, with fewer nerve endings.
DermNet NZ is a popular and trusted online dermatology repository, edited by the New Zealand dermatologist Amanda Oakley and used by doctors the world over. Its entry on “ethnic dermatology” claims that “darker skin tends to have a thicker dermis”, even though this notion has been debunked. (After I raised this with Oakley, she agreed to have this article revised. On request, she also pledged to include more pictures of darker skin tones with every DermNet NZ article.)
It is a positive step that we now have more specialists with experience of darker skin types, but these clinics come with their own potential pitfalls. Many seem to focus on minor, cosmetic skin concerns – often skin-lightening “treatments” – and nearly all the clinics in the UK are run privately, for profit. What started with Kenney offering a reparative service, helping people of colour to receive the same quality of care as white people, can easily become corrupted under the influence of the market. The danger is that such clinics become part of a lucrative industry that preys upon people who feel their skin is flawed because it is dark. If we are not careful, we could end up with a branch of medicine that medicalises blackness and then profits from the very patients the industry makes feel abnormal.
Skin conditions are more than just a nuisance – they can kill. Bob Marley’s early death from skin cancer is totemic, and not just because he was a musical giant. His story reminds us of the extra complications that black patients have to navigate when seeking healthcare, and how our health systems are set up in a way that has improved treatment for white patients while leaving people of colour behind.
Marley first told his friends that something was wrong in the summer of 1977. During a game of football in Paris, he injured his right big toe, and his toenail became painful. He admitted it was not the first time – he’d had a spot under his nail for a few years, and assumed it was a tiny bruise. He had to see two doctors before he was offered a biopsy, which confirmed the deadliest kind of skin cancer: acral lentiginous melanoma. Unlike the other three kinds of melanoma, which usually occur on soft, sun-exposed skin, acral lentiginous melanoma occurs in easy-to-miss places, such as the soles of your feet, or under your toenails.
The kind of melanoma that Marley developed, and which is by far the most common subtype in dark-skinned people, was not on the radar of most doctors. In 1977, the year Marley first developed symptoms, the world’s most popular medical textbook, the Merck Manual of Diagnosis and Therapy, published its 13th edition. It has entries on the three subtypes of melanoma that are most common in pale skin, but acral lentiginous melanoma isn’t even mentioned. Even today, it is the subtype of melanoma with the fewest successful treatment options.
Sunlight damages skin DNA, and while this is normally corrected within seconds, sometimes these genetic errors cannot be fixed, causing cancer. That’s why white people, who have less melanin to shield their DNA, are more than 20 times more likely to get skin cancer than black patients. But what’s really striking is how, in the US, the five-year survival rate after a melanoma diagnosis has changed in these two sub-populations, from the 70s to the present day. In white patients, the survival rate has improved from 68% to 90%. In black patients, it has gone down from 67% to 66%.
Such health outcome disparities help explain why a 2007 study in the American Journal of Public Health found that black patients were less likely than white people to trust medical professionals. They have good reason to be sceptical, given medical science’s long history of racism. It was only in 1975, for instance, that the family of Henrietta Lacks became aware that tissue removed from her body in 1951, without consent, was still being used in lucrative medical research. Then there is the case of the Tuskegee Study, a shocking medical experiment conducted in the US between 1932 and 1972, in which black men were denied treatment for syphilis so that investigators could study exactly how the disease killed people.
As a result of these betrayals, and an often condescending attitude from doctors – I have heard doctors bemoan the fact that black patients come too late to be cured, or refuse to engage with cancer screening programmes, or hold unscientific beliefs that hold them back – many black people have stopped entrusting their bodies to medicine altogether, a phenomenon sociologists have termed “the wisdom of distrust”. This forces some to seek help from alternative practitioners, or avoid treatment entirely. Marley himself missed some follow-up appointments with his doctors, instead trying an unproven dietary regime recommended by Josef Issels, a German quack.
By the time Marley was eventually given a skin graft, his cancer had already grown down through the layers of his skin and started its spread. In September 1980, he played his final show, before trying some last-ditch conventional cancer treatment. It didn’t work. Weighing just 37kg, he died on 11 May 1981 at the age of 36, at the Cedars of Lebanon Hospital in Miami.
A year after Marley died, the state of Florida started collecting cancer data in a state-wide registry. Shasa Hu, a melanoma expert and researcher at the University of Miami Hospital (the same hospital in which Marley died, now renamed), has studied the data on melanoma outcome disparities between white patients and patients of colour. Her research has shown how little has changed in the past four decades; just like in the 80s, black patients in Miami are still far less likely to be diagnosed in time to be cured.
I am not a dermatologist, but as a primary care physician, I am the first person people bring their rashes to, and nearly all doctors examine skin several times every day. But our training is woefully lacking, leaving medics entirely unprepared to serve our patients of colour.
There is a growing movement online to change this. In September 2019, unable to find adequate information regarding a rash her mixed-race son had, Ellen Buchanan Weiss set up the website Brown Skin Matters. It provides concerned parents and doctors with a collection of images showing how skin conditions both common and rare can present differently in richly pigmented skin. And as I write, there is a petition urging the UK General Medical Council to require that medical schools include a diverse representation of skin tones in their teaching.
One of the youngest people campaigning for this is Malone Mukwende, a second-year medical student at St George’s Medical School in London, who has co-authored a handbook, Mind the Gap, which aims to help fill this educational blind spot. But, while important, diversifying the skin tones we learn from is just the first, and easiest, step in a much broader struggle to reshape the medical profession to make it foreground the perspectives of people of colour, and eliminate the disparities that they have long suffered.
Despite having more than a decade of experience as a doctor, I have already learned a lot by going through the Brown Skin Matters website – which is good, not only for my future patients, but also for me. I soon realised that, despite experience in examining skin, I had never seen my own skin that way; I was always the reader, but never the page. I was shocked to see that several of the images showed skin changes that I have had myself, but which I had never thought to diagnose: the dry, crazy-paving skin on my lower legs looks like their picture of icthyosis; and I sometimes get rough patches that would do for nummular eczema, which can look subtle on brown skin.
Even right now, I have an unusual, itchy rash on my ankles. I’ve heard of similar rashes in people who have Covid-19, and it can be the only symptom. But again, I cannot find the information to know for certain. Jenna Lester, an assistant professor of dermatology at the University of California San Francisco, recently led a systematic review of papers describing Covid-19 skin changes. She found that, of the 130 images she reviewed, 92% showed white skin, 6% showed medium-brown skin, and none showed dark brown or black skin. So I have not even mentioned my rash to my own doctor. Because I know that they, like me, won’t know what to do. For now, we are all operating in the dark.














